Electricity: Basic Principles
Matter - anything that occupies space and has weight.
Element - a substance that cannot be decomposed any further by chemical action.
Compound - a combination of two or more elements.
Molecule - smallest particle that a compound can be reduced to before it breaks down into elements.
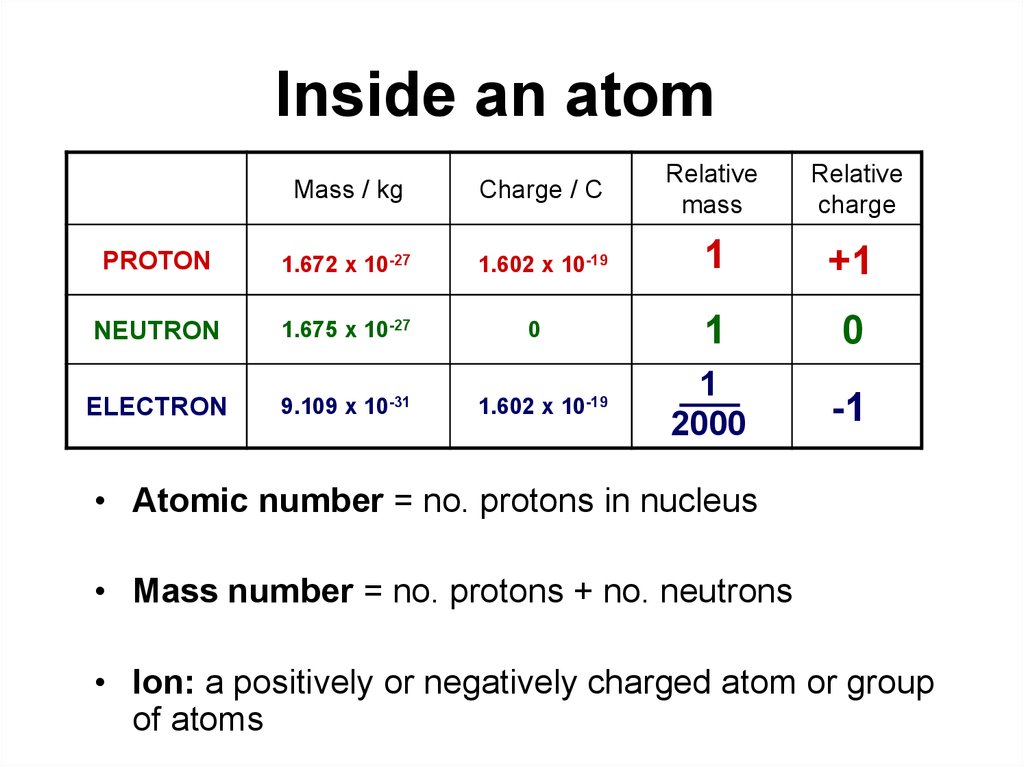
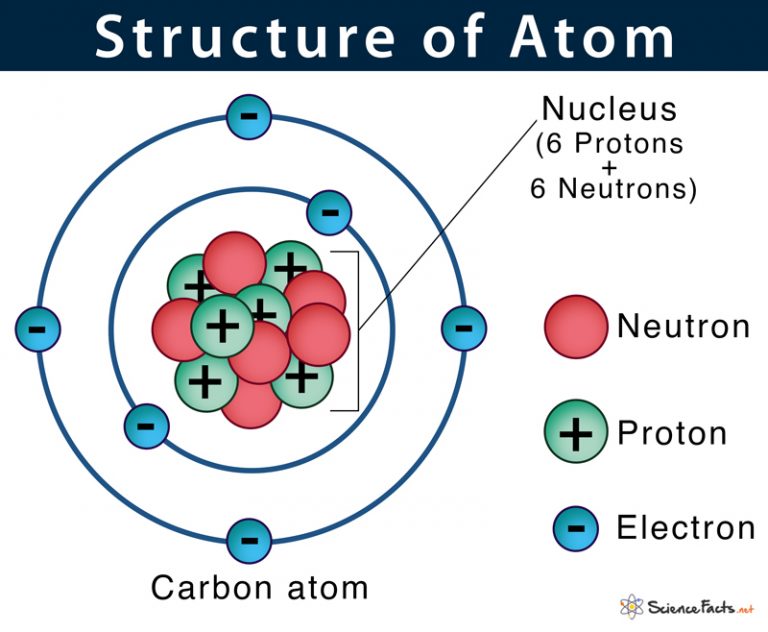
Atom - smallest part that an element can be reduced to and still be keeping the properties of the elements.
Parts of an Atom:
Elements - No. of electrons - No. of protons - No. of neutrons - No. of valence electron(s)
---------------------------------------
Copper - 29 - 29 - 34 - 1
Aluminum - 13 - 13 - 14 - 3
Germanium - 32 - 32 - 41 - 4
Electric Charge
A body is said to be charge, if it has either excess or deficit of electrons from its normal values due to sharing.
Coulomb (C) - unit of electric charge, which is equivalent to 6.25x10^18 electrons or protons. Named after the French physicist, Charles A. Coulomb (1736-1806).
Note: 1 C = 6.25x10^18 e
Potential Difference
Potential - the capability of doing work.
Any charge has the capability of doing work of moving another charge either by attraction or repulsion.
a. 6 electrons will be attracted by the +2 C plate and 3 electrons will be attracted by the +1 C plate, making a resultant motion of 3 electrons going towards the +2 C plate.
b. 3 electrons will be attracted by the +1 C plate and 3 electrons will be repelled by the -1 C plate, making a resultant motion of 6 electrons going towards the +1 C plate.
The net number of electrons moved in the direction of the positive charge plate depends upon the potential difference between the two charges.
Volt (V) - unit of potential difference, which is equal to one joule of work done per one coulomb of charge. Named after Italian physicist, Alessandro C. Volta (1754-1827) who invented the first electric battery.
Electric Current
When a potential difference between two charges forces a third charge to move, the charge in motion is called an electric current.
Ohm (Ω) - practical unit of resistance, Named after the German physicist, Georg S. Ohm (1787-1854).
Formula: R = ρL/A, R = ρV/A², R = ρL²/V
where:
R = resistance (ohm)
A = cross-sectional area (square meter)
ρ = resistivity (ohm-meter)
L = length (meter)
V = volume (cubic meter)
Specific resistance (resistivity) - resistance offered by a unit cube of the material.
Circular mil (CM) - area of a circle having a diameter (d) of one mil.
Conversion factor:
CM = d²
1000 mil = 1 inch
1 MCM = 1000 CM
Effect of Temperature in Resistance
Experiments have shown that the resistance of all wires generally used in practice in electrical systems, increases as the temperature increases.
Formula:
R₁/R₂ = (T+t₁)/ (T+t ₂), R₂/R₁ = 1 + α∆t
α = 1 / (T+t₁), ∆t = t₂ - t₁
where:
R₁ = initial resistance (ohm)
R₂ = final resistance (ohm)
T = inferred absolute temperature, temperature when resistance of a given material is zero.
t₁ = initial temperature